Oxford AQA International A Level Chemistry answers.
Explanation:. For a compound to be considered aromatic, it must be flat, cyclic, and conjugated and it must obey Huckel's rule. Huckel's rule states that an aromatic compound must have pi electrons in the overlapping p orbitals in order to be aromatic (n in this formula represents any integer). Only compounds with 2, 6, 10, 14.
The Chemistry department at Aston aims to inspire and enthuse its students in the study of chemistry. To do this, the department focuses primarily on the practical side of chemistry, whilst trying to encourage students to think deeply about and understand the underlying principles and big ideas of chemistry as a science. Staff: Mr R Giubertoni, Head of Department; Mr S Hall, Teacher; Dr R.
Sexual health help and advice; Mental health information; Long-distance relationships; Guide to contraception; Common student health problems; Useful welfare phone numbers; Dealing with a break-up; Student finance. Undergraduate. Full time; Part time; EU Students. Full time; Part time; Taking a break or withdrawing from your course; Postgraduate. Postgraduate Master’s Loan; Postgraduate.
This book provides an up-to-date and comprehensive account of aromatic chemistry. A series of chapters describes the synthesis and reactions of the major functional derivatives of benzene and the more common polycyclic systems. The concepts of aromaticity and the mechanism of aromatic substitution are discussed, as is the use of metals in the synthesis of aromatic compounds. Throughout.
Aromatic Chemistry. STUDY. PLAY. What is Kekule s structure of benzine? a ring of carbon atoms, where there is alternating single, double bonds. also known as cyclohex-1,3,5-ene. Explain one reason the Kekule structure of benzene was discredited. - The enthalpy of hydrogenation was found to be much less exothermic than expected. No double bonds so less energy is released than expected. More.
The distinctive electronic structure of aromatic leads to some distinctive reactivity! We will be covering the naming of benzene derivatives, stability of aromatic compounds, electrophilic aromatic substitution, and nucleophilic aromatic substitution.
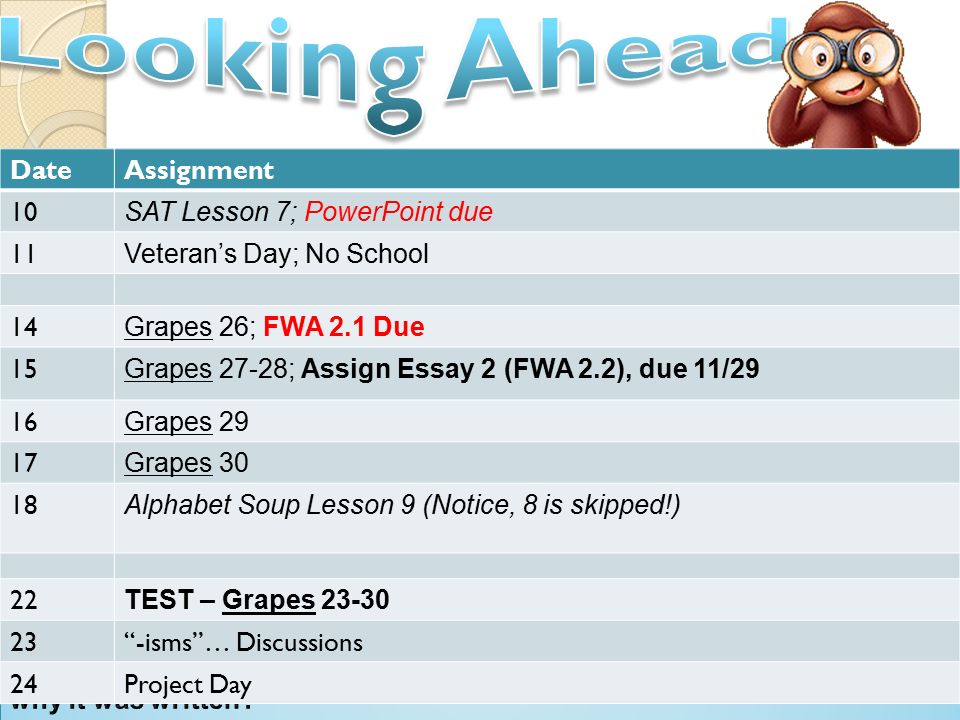
Throughout this course you will be assessed in the following ways; - Through completion of weekly homework - End of topic assessments - Formal assessments - End of year externally marked exam papers. Year 1 - 2 papers - Breadth in Chemistry - Depth in Chemistry Year 2 - 3 papers - Periodic table, elements and physical Chemistry - Synthesis and analytical techniques - Unified Chemistry.